Posted: October 4, 2019
Researchers in the college are studying the biology of insect vectors of disease with a goal of preventing outbreaks.
Even the most gentle among us would slap away a mosquito to avoid being bitten; indeed, most would agree that these insects are a nuisance. But beyond being annoying these insects can also transmit deadly diseases.
In tropical areas, the problem is particularly devastating. More than 700,000 people die each year from vectorborne illnesses--such as malaria, dengue fever, yellow fever, and Japanese encephalitis--according to the Centers for Disease Control and Prevention. Malaria alone kills 400,000 people each year--most of them children under the age of 5.
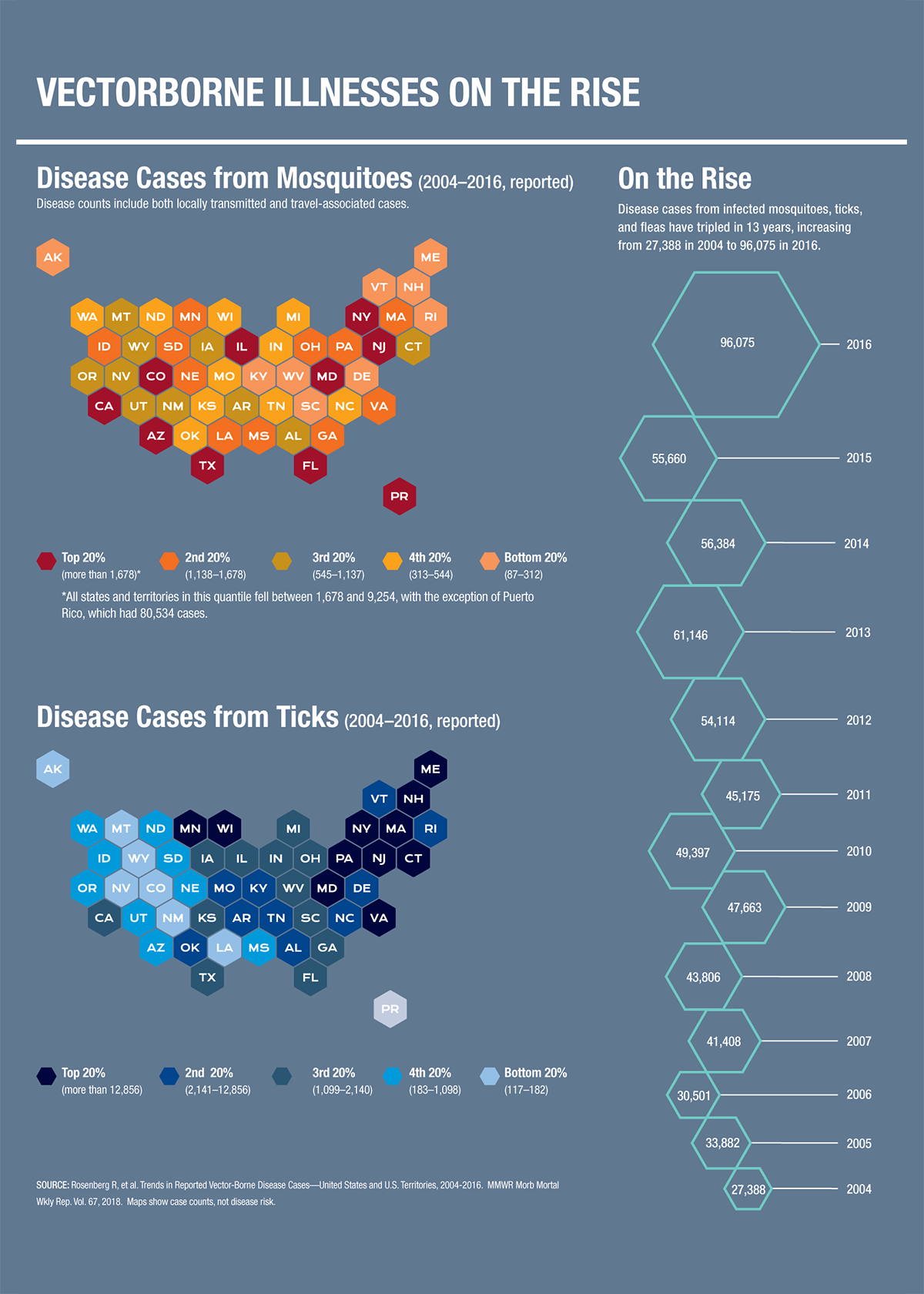
In the U.S. we aren't as safe as we might think. Here, vectorborne diseases are on the rise. In fact, cases tripled from 2004 to 2016, according to the World Health Organization. During that period, Lyme disease accounted for 82 percent of all tickborne disease, and mosquitoes transmitted numerous viruses--including Zika and chikungunya--in Puerto Rico, the U.S. Virgin Islands, and American Samoa. On the mainland, West Nile virus periodically hit epidemic proportions. And with global climate change, these diseases and others could become more prevalent.
Researchers in the college are studying the biology of insect vectors to understand what it is about them that makes them good carriers of disease and how they might be controlled. "Research in the college takes an integrated approach to understanding how animals, humans, plants, and insects respond to environmental pressures such as vectorborne diseases," says Gary Thompson, associate dean for research and graduate education. "This is critical especially as the world population continues to grow and the necessary increased animal and plant production creates conditions favorable to the spread of diseases or even the emergence of new pathogens."
According to Andrew Read, director of the Huck Institutes of the Life Sciences, Evan Pugh Professor of Biology and Entomology, and Eberly Professor of Biotechnology, the college is particularly well positioned to conduct such work. "Penn State has one of the best entomology programs in the world, and one of the strongest groups in infectious disease ecology and evolution," he says. "Vectorborne disease is right at the intersection of those two topics. Recognizing this, the University--via the Huck Institutes of the Life Sciences and the College of Agricultural Sciences--has invested in state-of-the-art facilities for this sort of work and continues to hire the best scientists."
Who's Who

Andrew Read
According to Read, many people consider mosquitoes to be the most dangerous animal in the world due to the large number of diseases they transmit. Identifying which mosquito species harbor which diseases is an important first step in studying disease transmission. For example, worldwide, some 3,500 species of mosquitoes exist, but only a few species in the genus Anopheles transmit the malaria virus. And Zika and chikungunya viruses are carried by just two species: Aedes aegypti and Aedes albopictus.
Jason Rasgon, professor of entomology and disease epidemiology, has conducted numerous studies to identify the vectors of certain diseases. Lately, he is particularly interested in Mayaro virus, which was first described in 1954 in Mayaro County, Trinidad. In humans, the virus causes rash, fever, myalgia, eye pain, headache, diarrhea, and joint pain that can persist for months or even years. Over the years, sporadic outbreaks of the virus have cropped up in South America and the Caribbean. Recently, however, several imported cases of Mayaro virus have been reported in the Netherlands, Germany, France, and Switzerland. "These cases highlight the need to survey such regions, including the United States, for possible introductions of this potentially dangerous virus," says Rasgon.

Jason Rasgon
Along with his colleagues, Rasgon is investigating the species of mosquito that transmit Mayaro virus. "Aedes aegypti was thought to be the main vector of the virus, but it's actually not that good," he says. "Our research shows that Anopheles mosquitoes are much more efficient vectors of Mayaro virus."
Every species of Anopheles mosquito that Rasgon's team tested, including those found in the southeastern United States and even up into Pennsylvania, demonstrated good ability to carry and transmit the virus. "We're starting to really look at Anopheles as drivers of Mayaro and potentially other arboviruses [viruses transmitted by mosquitoes] because it's really been a neglected area of study," says Rasgon. He and his colleagues have done similar work for Zika to determine which potential vectors in North America can transmit that virus, which causes severe birth defects.
Temperature Dependence

Matthew Thomas
In addition to identifying which mosquito species carry which diseases, researchers in the college are studying the environmental conditions that are needed for mosquitoes to thrive.
Matthew Thomas, professor and Huck Scholar in Ecological Entomology, is particularly interested in temperature. "Mosquito biting rates, longevity, how many eggs they lay, and how quickly the pathogens they transmit develop are all strongly influenced by temperature," he says. "It's a bit like Goldilocks; each species of mosquito and pathogen has an optimum temperature range that is just right."
Thomas and his colleagues are working to determine the "just-right" temperature ranges for a number of disease-causing mosquito species. Their goals include gaining a greater understanding of mosquito biology and predicting the insects' responses to changing environments.
According to Thomas, a one- or two-degree change in temperature could cause a dramatic shift in risk of diseases such as malaria
"Huge populations of people live at higher elevations in parts of East Africa, quite possibly because temperatures are cool and malaria transmission is low," says Thomas. "If the climate warms, suddenly you've got many more people at risk of malaria."
In the lab, Thomas has exposed different species of mosquitoes to a range of temperature and other environmental conditions, with a goal of identifying their lower, optimum, and upper temperature limits. The information will help scientists predict how mosquitoes will react to not only climate change but also potentially more immediate changes in the built environment.
"There is a very dramatic change in housing happening across Africa as householders switch from using traditional building materials," says Thomas. "Putting a metal roof on your house can lead to a substantial change in the temperatures experienced by mosquitoes as they feed and rest indoors. This is something that can happen overnight, without waiting for climate change."
It will also help researchers better understand the consequences of mosquito adaptation to certain control tools. For example, Thomas says the introduction of insecticide-laced bed nets has led to a marked reduction in malaria transmission. These nets work because they block mosquito feeding as people sleep at night. However, there is now evidence that some mosquitoes are shifting their feeding time to the evening or the morning so they will be more likely to access humans outside of the protection of their bed nets. However, the ambient temperature at these times is different and could affect the mosquitoes' abilities to harbor the malaria parasite.
"The initial stage of parasite development right after a mosquito takes in a blood meal is very sensitive to temperatures above 30 degrees Celsius," says Thomas. "What we don't yet know is whether biting in the early evening or the early morning will alter the ability of parasites to infect mosquitoes, but this is key to understanding the epidemiological impact of mosquito behavioral resistance."
A Bacterial Solution

Elizabeth McGraw
Other factors also influence a mosquito's ability to transmit disease. Elizabeth McGraw, professor and Huck Scholar in Entomology, is studying one of them--a bacterium that lives inside the cells of mosquitoes.
"Wolbachia lives inside about 50 percent of all insects," says McGraw. "Its presence can prevent the establishment of the virus that causes dengue fever."
According to McGraw, Wolbachia prevents uninfected females in populations from producing successful offspring. "It means that infected females by default have an advantage because their offspring are more likely to populate the next generation, so Wolbachia spreads; each generation it rises in frequency," she says.
Recognizing this phenomenon, scientists have attempted to use the bacterium to control the spread of disease. They have injected Wolbachia into Aedes aegypti, the mosquito species that causes dengue fever, chikungunya, Zika fever, Mayaro virus, and yellow fever virus. "What they found was when Wolbachia was present in the mosquito it reduced the ability of the mosquito to transmit virus; it was like vaccinating the mosquito," says McGraw.
Currently, that finding forms the basis of a very large multi-million-dollar effort to release these mosquitoes throughout the tropics. McGraw helped with the initial research and release of these mosquitoes in Australia. A large nonprofit organization is now working to introduce them in Indonesia, Brazil, Colombia, and other places. "The goal is to release these mosquitoes into wild populations and get them to spread and take over so they replace all the mosquitoes that are there that don't have Wolbachia," says McGraw.
In addition to working on the original it releases in Australia, McGraw and her colleagues are conducting research into the biological mechanism of how Wolbachia reduces virus infection in mosquitoes. A variety of hypotheses have been posed, she says, including competition for space between the virus and the bacterium in the mosquito's cells, and an immune response to Wolbachia that makes it harder for the virus to get a toehold.
"The reason we need to understand the mechanism is because these bacteria are being introduced into populations all over the world and at some point the viruses are going to evolve a way to sneak around the Wolbachia," she says. "To prepare for that, we need to understand how the whole thing works."
But, she cautions, using Wolbachia will not be a silver bullet. "We're still going to need insecticides, vaccines, and management of mosquito breeding sites," she says. "The more strategies we have in place, the greater our success against dengue and other viruses."
Genetic Design
One of those stratgies is direct genetic manipulation of mosquitoes. Jason Rasgon and his colleagues are working to improve a relatively new gene-editing technology, called CRISPR/Cas9, for use in mosquitoes. When fine-tuned, the technique will enable researchers to genetically modify mosquitoes so they don't spread disease.
Rasgon explains that genetic engineering in any organism normally begins with injecting DNA and gene editing material into eggs using a fine needle. "With mosquitoes, the eggs often do not take well to being injected," he says. "We've developed instead a way to directly inject the mother, who then passes the DNA modifications on to her offspring."
Working with McGraw, Rasgon is using his technique to edit the genomes of mosquitoes so they are resistant to viruses. "My lab is studying the immune reactions of mosquitoes and what genes are involved in their own natural abilities to control viruses," says McGraw. "We're looking for natural variation in mosquito populations. We give Jason our top candidates, then he uses his technology that he's developed to make genetic modifications in mosquitoes so they, too, exhibit immune reactions to viruses."
Rasgon is using the CRISPR/Cas9 technology to alter mosquito genomes in other ways as well, and he has even branched out to other organisms, including ticks and other insects and arthropods.
"Ultimately, our goal is to understand the biology of disease-causing organisms and prevent them from harming human health," he says. "When people don't have to spend their lives worrying about getting sick or their children dying, they are free to focus on other things that are important to society."
Read adds, "My hope is that we will look back on our careers at Penn State and see that lives were saved by what we did here."
-- By Sara LaJeunesse
Features
Breaking the Silence on Farm Stress
Farming has always been a demanding profession, but today's farmers face unprecedented pressures that can severely impact their mental health.
Biting Back
Research Targets Vector-Borne Diseases to Save Lives
Leading Forward
Ott brings deep connection to role of dean.